Fertility
Corina™Intrauterine Estrogen-Eluting Stent
 Sustained-Release of 17-β Estradiol (0.2 mg/day)
Physical Barrier to Prevent Adhesion Formation
Stable estrogen release for 60 days,Advance endometrial recovery.
Sustained-Release of 17-β Estradiol (0.2 mg/day)
Physical Barrier to Prevent Adhesion Formation
Stable estrogen release for 60 days,Advance endometrial recovery.
Clinical data demonstrate highly statistically significant superiority. ( P <0.001)
Soft, safe, and non-compressive.
The test group showed significantly better adhesion reduction than the control group at 60 days (P<0.001).
Intrauterine Adhesions (IUA)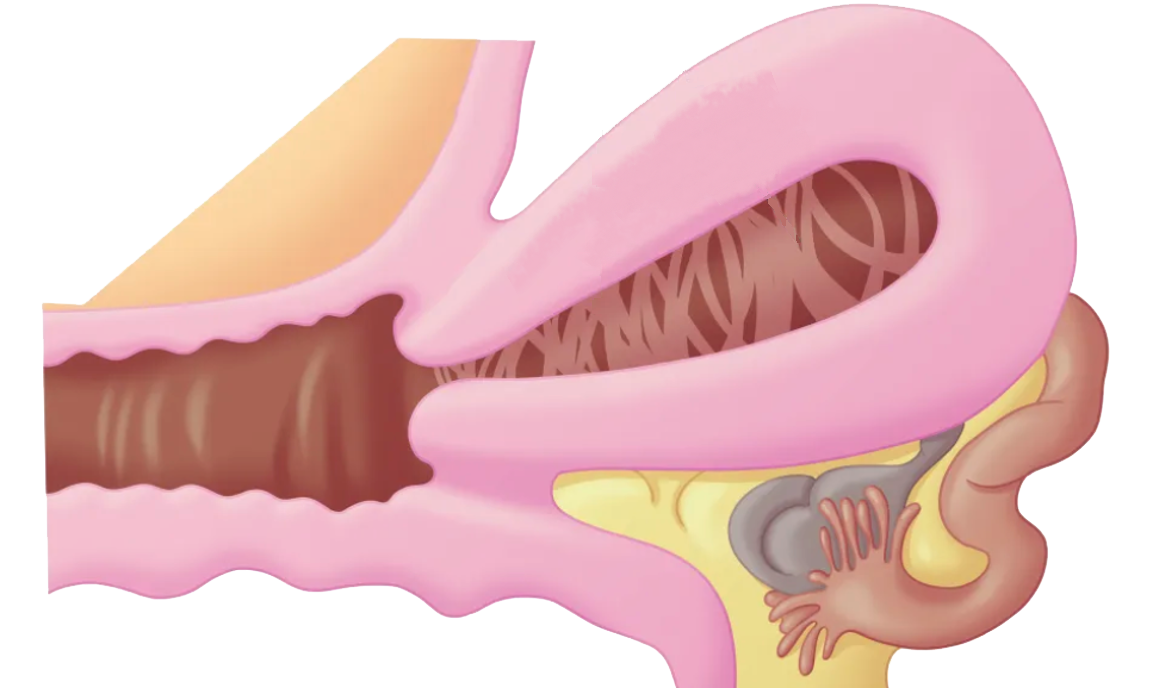 Intrauterine Adhesions (IUA), also known as Asherman’s syndrome, occur when damage to the basal layer of the endometrium leads to endometrial fibrosis, intrauterine scarring, and the formation of adhesions that may partially or completely occlude the uterine cavity [1–2].
Intrauterine Adhesions (IUA), also known as Asherman’s syndrome, occur when damage to the basal layer of the endometrium leads to endometrial fibrosis, intrauterine scarring, and the formation of adhesions that may partially or completely occlude the uterine cavity [1–2].
Clinically, IUA may cause menstrual abnormalities (e.g., hypomenorrhea or amenorrhea) and cyclic pelvic pain. It is also associated with infertility, recurrent pregnancy loss, preterm delivery, abnormal placentation (e.g., placenta accreta), and ectopic pregnancy. The severity of adhesions is directly correlated with reproductive prognosis.
Routes of Estrogen Administration Estrogen preparations differ substantially in bioavailability, duration of action, and safety profiles. These differences determine the choice of administration route, dosing strategy, and overall treatment protocol. Routes of Estrogen Administration Intrauterine Oral Transdermal Estrogen promotes endometrial repair by upregulating epithelial regeneration, facilitating mesenchymal-to-epithelial transition, and stimulating vascular endothelial growth factor (VEGF) expression, thereby supporting both structural and functional recovery of the endometrium.
What is Corina? Corina™ is an intrauterine estrogen-releasing system designed to deliver estrogen locally and continuously to the uterine cavity, supporting endometrial regeneration while minimizing systemic exposure. Clinical evidence-Clinical trial To compare the efficacy and safety of the silicone rubber estrogen-releasing intrauterine system versus Foley balloon catheter combined with intrauterine cross-linked sodium hyaluronate gel combined Research purpose: Research design: This was a multicenter prospective randomized controlled 2-arm parallel trial . Sample size: The trial design was a superiority test with planned 246 cases and actual 250 cases enrolled as 1:1 in the test and control groups. Clinical evidence-Trial design Dydrogesterone 20mg/d for 5 days after stent removal Screened subjects Transcervical resection of adhesions (TCRA) 6mg/d estradiol valerate for 60 days, additional 10mg/d of dydrogesterone for the last 10 days Test group: With Corina™ intrauterine drug-eluting stent placed in the uterine cavity postoperatively Control group: With Foley balloon + self-crosslinking sodium hyaluronate gel placed and postoperative estrogen-progestin cycle therapy assisted postoperatively Follow-up on postoperative menstrual improvement, laboratory examinations*, abnormal postoperative bleeding, pain conditions, and reproductive tract infections Back to hospital for a second inspection 2 months postoperatively to remove the uterine stent and re-evaluate the intrauterine conditions Postoperative follow-up at 12 months to assess pregnancy and pregnancy outcome, number of TCRAs and adverse events
![]()
 Test group n=125
Test group n=125
 +
Control group n=125
+
Control group n=125
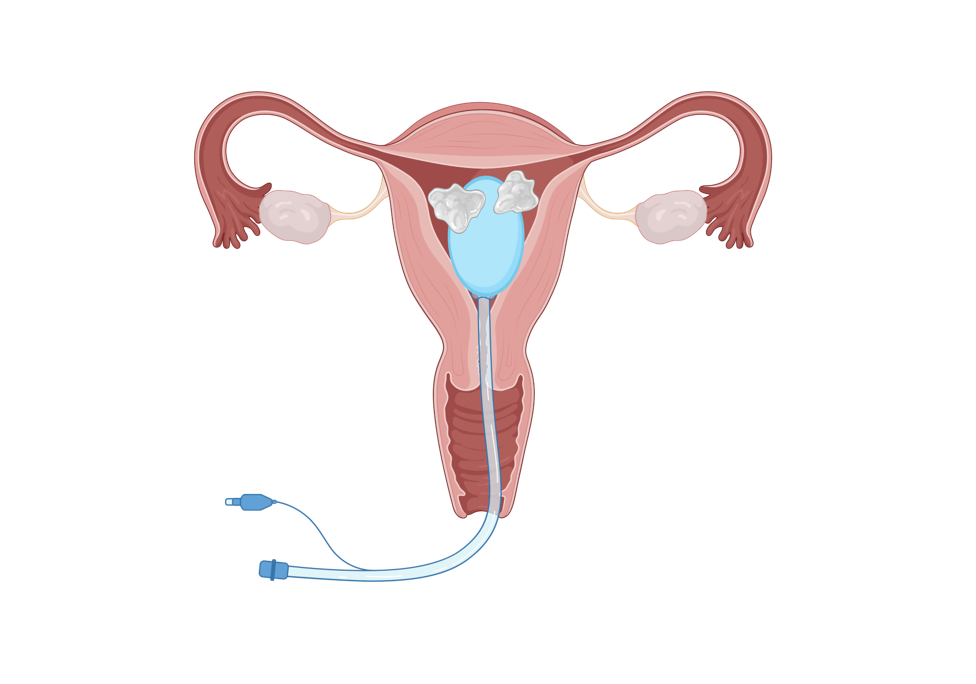
 Clinical evidence-Clinical trial
93.33%
58.56%
0
20
40
60
80
100
Unit:%
Corina™
Foley Balloon
Clinical evidence-Clinical trial
93.33%
58.56%
0
20
40
60
80
100
Unit:%
Corina™
Foley Balloon
+ Hyaluronic Acid Gel
+ Oral Estrogen Pill 4.31 + 7.46 mm 0.75 ± 2.73 mm 0.0 1.0 2.0 3.0 4.0 5.0 Unit:mm Corina™ Foley Balloon
+ Hyaluronic Acid Gel
+ Oral Estrogen Pill Baseline Endometrial Thickness
Corina™: 4.32±1.73 mm
Control Group:4.07±1.46 mm [1]Feng et al. BMC Medicine (2024) 22:395https://doi.org/10.1186/s12916-024-03608-4
[2]Chinese Journal of Reproduction and Contraception, September 2025, Vol. 45, No.9
[3] Dreisler E, Kjer JJ. Asherman‘s syndrome: current perspectives on diagnosis and management[J]. Int JWomens Health, 2019, 11: 191-198. DOI:10.2147/IJWH.S165474.
[4] Wang PH, Yang ST , Chang WH, et al. Intrauterine adhesion[J].Taiwan J Obstet Gynecol, 2024, 63(3): 312-319. DOI:10.1016/j.tjog.2024.02.004. Conclusions:
The efficiency of adhesion reduction was significantly better in the test group than that in the control group 60 days postoperatively. (P<0.001) Corina- operation demonstration Corina™Intrauterine Estrogen-Eluting Stent Intrauterine Delivery : Safety. Precision. Efficacy
 Intrauterine adhesions (IUA) result from damage to the basal layer of the endometrium, leading to fibrosis, scar formation, and partial or complete obliteration of the uterine cavity, ultimately impairing reproductive outcomes.
Intrauterine adhesions (IUA) result from damage to the basal layer of the endometrium, leading to fibrosis, scar formation, and partial or complete obliteration of the uterine cavity, ultimately impairing reproductive outcomes.
The primary goal of treatment is to restore the normal uterine cavity anatomy and promote endometrial regeneration. Comprehensive postoperative management is essential to enhance endometrial repair, inhibit fibrosis, prevent recurrence, and support timely fertility restoration in patients with reproductive intentions.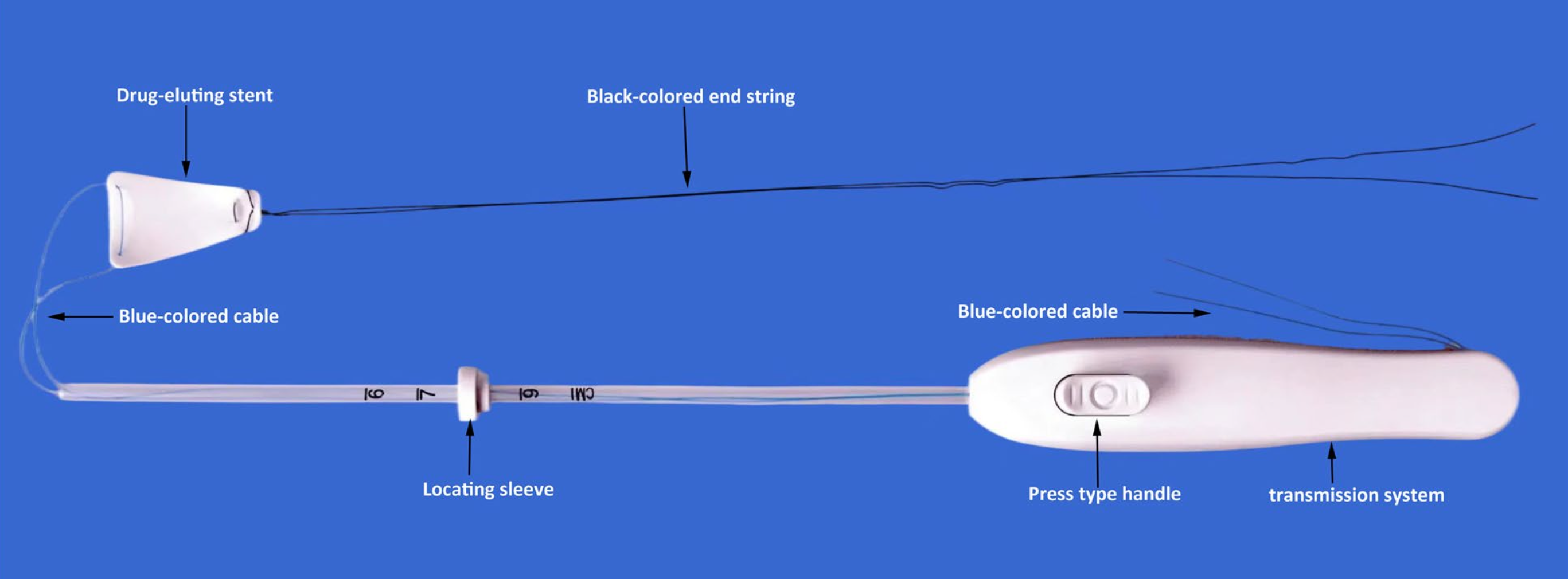 +
↓
Conclusions:
+
↓
Conclusions:
Endothelial thickness was significantly better in the test group than that in the control group at 60 days postoperatively (P<0.001).
The intrauterine estrogen-releasing system could be more effective at promoting endometrial proliferation in the test group than oral estrogen in the control group.
References ↓ ↓ ↓ ↓ ↓ P<0.001 P<0.001
 Sustained-Release of 17-β Estradiol (0.2 mg/day)
Physical Barrier to Prevent Adhesion Formation
Stable estrogen release for 60 days,Advance endometrial recovery.
Sustained-Release of 17-β Estradiol (0.2 mg/day)
Physical Barrier to Prevent Adhesion Formation
Stable estrogen release for 60 days,Advance endometrial recovery.Clinical data demonstrate highly statistically significant superiority. ( P <0.001)
Soft, safe, and non-compressive.
The test group showed significantly better adhesion reduction than the control group at 60 days (P<0.001).
Intrauterine Adhesions (IUA)
 Intrauterine Adhesions (IUA), also known as Asherman’s syndrome, occur when damage to the basal layer of the endometrium leads to endometrial fibrosis, intrauterine scarring, and the formation of adhesions that may partially or completely occlude the uterine cavity [1–2].
Intrauterine Adhesions (IUA), also known as Asherman’s syndrome, occur when damage to the basal layer of the endometrium leads to endometrial fibrosis, intrauterine scarring, and the formation of adhesions that may partially or completely occlude the uterine cavity [1–2].Clinically, IUA may cause menstrual abnormalities (e.g., hypomenorrhea or amenorrhea) and cyclic pelvic pain. It is also associated with infertility, recurrent pregnancy loss, preterm delivery, abnormal placentation (e.g., placenta accreta), and ectopic pregnancy. The severity of adhesions is directly correlated with reproductive prognosis.
Routes of Estrogen Administration Estrogen preparations differ substantially in bioavailability, duration of action, and safety profiles. These differences determine the choice of administration route, dosing strategy, and overall treatment protocol. Routes of Estrogen Administration Intrauterine Oral Transdermal Estrogen promotes endometrial repair by upregulating epithelial regeneration, facilitating mesenchymal-to-epithelial transition, and stimulating vascular endothelial growth factor (VEGF) expression, thereby supporting both structural and functional recovery of the endometrium.
What is Corina? Corina™ is an intrauterine estrogen-releasing system designed to deliver estrogen locally and continuously to the uterine cavity, supporting endometrial regeneration while minimizing systemic exposure. Clinical evidence-Clinical trial To compare the efficacy and safety of the silicone rubber estrogen-releasing intrauterine system versus Foley balloon catheter combined with intrauterine cross-linked sodium hyaluronate gel combined Research purpose: Research design: This was a multicenter prospective randomized controlled 2-arm parallel trial . Sample size: The trial design was a superiority test with planned 246 cases and actual 250 cases enrolled as 1:1 in the test and control groups. Clinical evidence-Trial design Dydrogesterone 20mg/d for 5 days after stent removal Screened subjects Transcervical resection of adhesions (TCRA) 6mg/d estradiol valerate for 60 days, additional 10mg/d of dydrogesterone for the last 10 days Test group: With Corina™ intrauterine drug-eluting stent placed in the uterine cavity postoperatively Control group: With Foley balloon + self-crosslinking sodium hyaluronate gel placed and postoperative estrogen-progestin cycle therapy assisted postoperatively Follow-up on postoperative menstrual improvement, laboratory examinations*, abnormal postoperative bleeding, pain conditions, and reproductive tract infections Back to hospital for a second inspection 2 months postoperatively to remove the uterine stent and re-evaluate the intrauterine conditions Postoperative follow-up at 12 months to assess pregnancy and pregnancy outcome, number of TCRAs and adverse events
 Test group n=125
Test group n=125
 +
Control group n=125
+
Control group n=125

 Clinical evidence-Clinical trial
93.33%
58.56%
0
20
40
60
80
100
Unit:%
Corina™
Foley Balloon
Clinical evidence-Clinical trial
93.33%
58.56%
0
20
40
60
80
100
Unit:%
Corina™
Foley Balloon+ Hyaluronic Acid Gel
+ Oral Estrogen Pill 4.31 + 7.46 mm 0.75 ± 2.73 mm 0.0 1.0 2.0 3.0 4.0 5.0 Unit:mm Corina™ Foley Balloon
+ Hyaluronic Acid Gel
+ Oral Estrogen Pill Baseline Endometrial Thickness
Corina™: 4.32±1.73 mm
Control Group:4.07±1.46 mm [1]Feng et al. BMC Medicine (2024) 22:395https://doi.org/10.1186/s12916-024-03608-4
[2]Chinese Journal of Reproduction and Contraception, September 2025, Vol. 45, No.9
[3] Dreisler E, Kjer JJ. Asherman‘s syndrome: current perspectives on diagnosis and management[J]. Int JWomens Health, 2019, 11: 191-198. DOI:10.2147/IJWH.S165474.
[4] Wang PH, Yang ST , Chang WH, et al. Intrauterine adhesion[J].Taiwan J Obstet Gynecol, 2024, 63(3): 312-319. DOI:10.1016/j.tjog.2024.02.004. Conclusions:
The efficiency of adhesion reduction was significantly better in the test group than that in the control group 60 days postoperatively. (P<0.001) Corina- operation demonstration Corina™Intrauterine Estrogen-Eluting Stent Intrauterine Delivery : Safety. Precision. Efficacy
 Intrauterine adhesions (IUA) result from damage to the basal layer of the endometrium, leading to fibrosis, scar formation, and partial or complete obliteration of the uterine cavity, ultimately impairing reproductive outcomes.
Intrauterine adhesions (IUA) result from damage to the basal layer of the endometrium, leading to fibrosis, scar formation, and partial or complete obliteration of the uterine cavity, ultimately impairing reproductive outcomes.The primary goal of treatment is to restore the normal uterine cavity anatomy and promote endometrial regeneration. Comprehensive postoperative management is essential to enhance endometrial repair, inhibit fibrosis, prevent recurrence, and support timely fertility restoration in patients with reproductive intentions.
 +
↓
Conclusions:
+
↓
Conclusions: Endothelial thickness was significantly better in the test group than that in the control group at 60 days postoperatively (P<0.001).
The intrauterine estrogen-releasing system could be more effective at promoting endometrial proliferation in the test group than oral estrogen in the control group.
References ↓ ↓ ↓ ↓ ↓ P<0.001 P<0.001


 enquiry@yipurunbio.com commercial@yipurunbio.com
Room 2206, No. 396, Lishizhen Road, Pudong New Area, Shanghai
enquiry@yipurunbio.com commercial@yipurunbio.com
Room 2206, No. 396, Lishizhen Road, Pudong New Area, Shanghai
